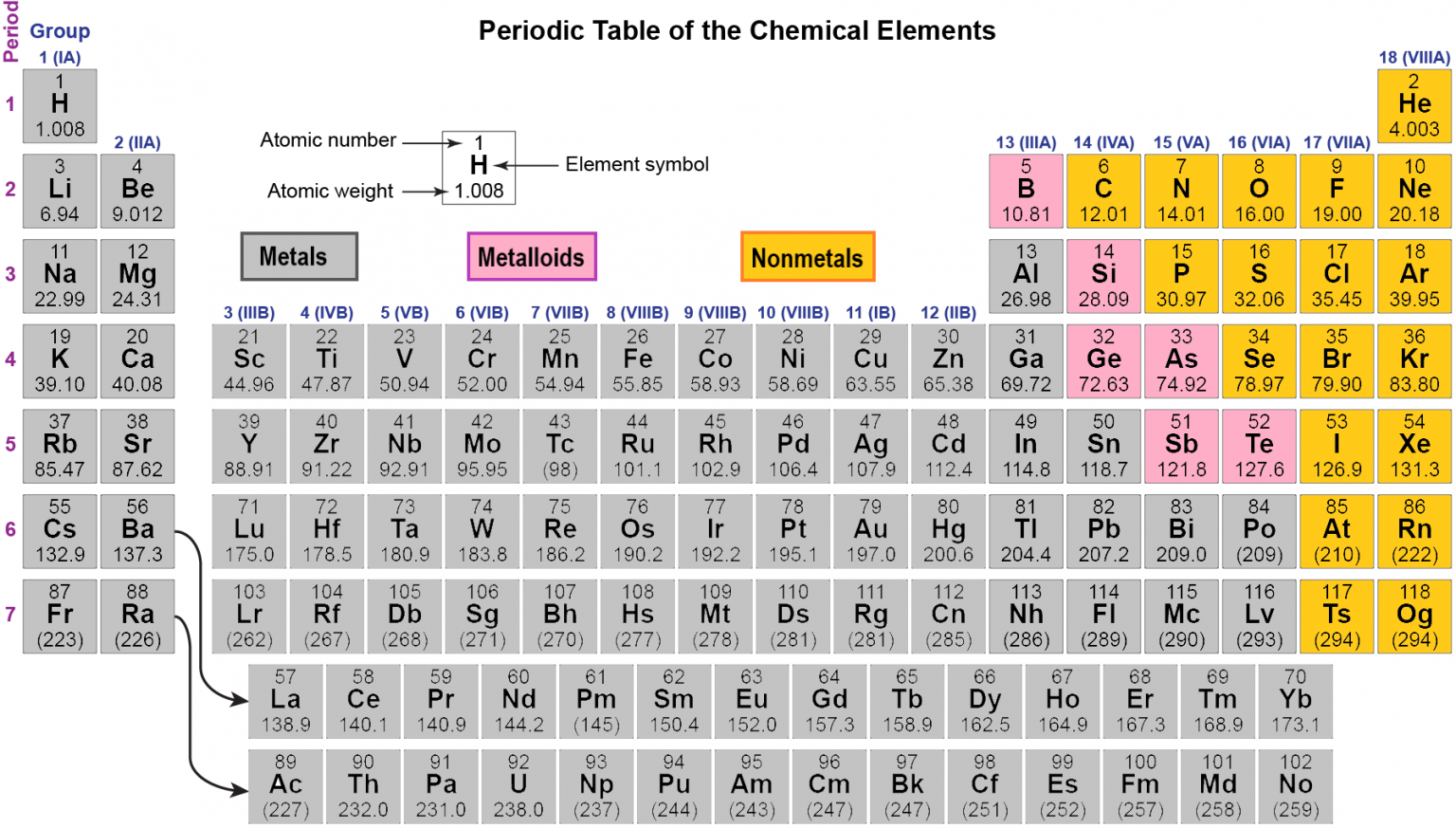
The groups will then present their findings to the class and discuss how they arrived at their findings.Īs a class we will group the elements under the three groups after discussing the criteria for assigning elements to groups. They will arrange the samples into metals, nonmetals and metalloids. They will discuss their findings in their own small groups and group the samples under two categories based on their physical and chemical properties and find which elements could fit into either of the groups and why. The student groups will then make a data table to compare the observations for the given samples. With copper (II) chloride which is a blue colored solution, they will be looking for color change of the liquid and of the sample. While there is no universally accepted standard, certain color schemes have become more widely used, like highlighting metals in yellow/orange, nonmetals in green, noble gases in purple, alkali metals in pink, alkaline earth metals in blue, and halogens in light green. To each sample, 15 to 20 drops of the acid solution will be added and they will look for any reaction (change ) happening like bubbles of hydrogen gas coming out. Today, many varieties of color-coded periodic tables exist. They will take a small portion of the given samples in a well plate. Then they will test for the reactivity of the samples with 0.5M hydrochloric acid and 0.1M copper (II) chloride solution. They will use the conductivity tester to see if the sample conducts electricity. They will use the hammer to test if the sample is brittle or malleable. They will look at the appearance of the given sample to see if it has luster or is dull. They are the most reactive of all metals. The first column of the periodic table, or group 1, is where the alkali metals are found. Metals are located to the left of the stairstep. All of them, except mercury, are solid at room temperature. The students have to group the samples under the categories of metals, nonmetals or metalloids. Atoms & the Periodic Table Metals are good conductors of heat and electricity.

The samples the student groups get are carbon (pencil lead works), pieces of magnesium ribbon, silicon lumps, sulfur lumps, iron filings, and pieces of mossy zinc. The students will form small lab groups of two or three members.Īfter I review the difference between physical and chemical properties, and the characteristics of metals, nonmetals and metalloids, the student groups will get samples in seven vials which have been coded with letters 'a' to 'g'.
